A supplement label tells you what a brand wants you to know. A certificate of analysis tells you what is actually in the product.
Most consumers never ask for one. Most brands are counting on that. The CoA is the single most useful document available for verifying whether a mushroom supplement contains what it claims to contain, and getting hold of it is simpler than most people think.
Mushroom Supplements: What Works, What’s Misleading, and How to Buy Safely
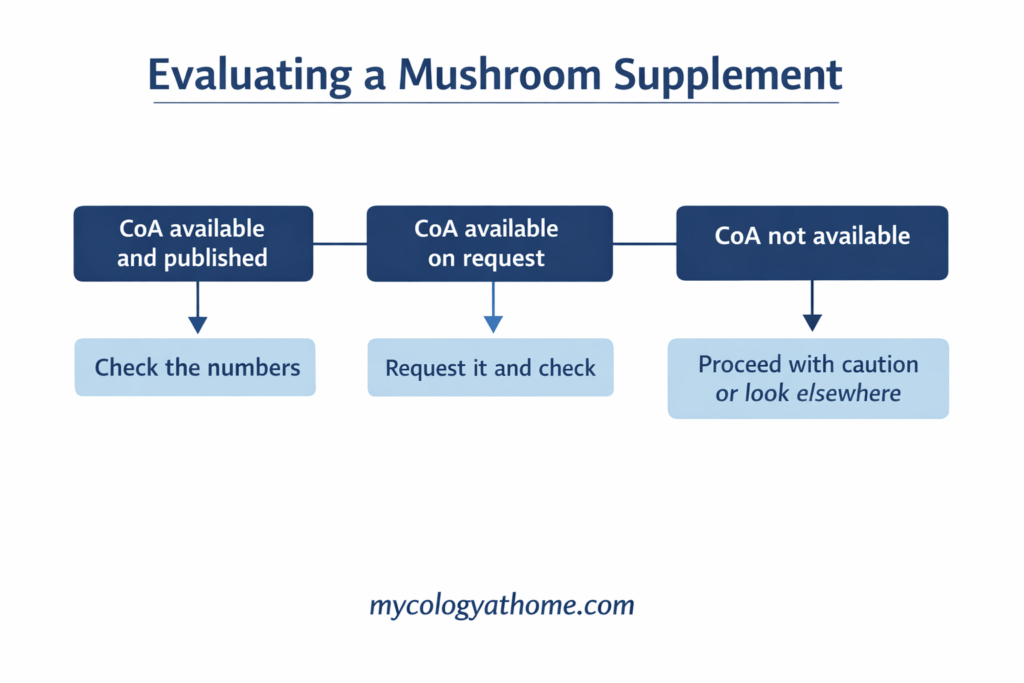
What a certificate of analysis actually is
A certificate of analysis is a document produced by a third-party laboratory that independently tests a specific batch of a product. It reports what the lab found when they analysed the material, rather than what the brand claims is in it.
For mushroom supplements, a CoA tests for active compound content, heavy metals, and microbial contamination, identified by batch number, test date, and the testing lab’s sign-off.
The key word is third-party. An internal quality document produced by the brand is not a CoA in any meaningful sense. A genuine CoA comes from a named, accredited facility with no commercial relationship to the brand.
Why it matters for mushroom supplements
Supplement labels in the United States do not require brands to disclose beta-glucan content, extraction method, or whether the product is fruiting body or mycelium on grain. A brand can list 1000mg of lion’s mane and legally leave out everything that would tell you whether that 1000mg is worth anything.
The CoA fills that gap. It shows you the actual measured beta-glucan percentage from an independent lab, which is the most important single number for evaluating whether a mushroom supplement is likely to deliver what the research supports.
Without a CoA, you are relying on a brand’s self-reported claims alone.
Mycelium on Grain: The Problem With Most Mushroom Supplements

What to look for on a CoA
Not all CoAs are equally useful. Here is what a good one includes.
Beta-glucan percentage
This is the primary quality indicator for most medicinal mushrooms. A quality fruiting body extract should test at 25 percent or higher. Mycelium on grain products often test at 1 to 5 percent. The CoA gives you an independently verified number rather than a self-reported label claim.
What Are Beta-Glucans? The Compound Behind Mushroom Supplements
Heavy metals testing
Mushrooms absorb compounds from their growing substrate, including heavy metals. The CoA should show results for lead, arsenic, cadmium, and mercury with pass or fail against established limits.
Microbial contamination
This covers bacteria, yeast, and mould counts. A CoA that includes microbial testing confirms the product was independently checked before sale.
Batch number and test date
A batch number ties the document to a specific production run. Without it you cannot confirm the CoA applies to what you are buying. A test date that is several years old is not useful for evaluating a current batch.
Lab name and accreditation
The lab should be named and independently verifiable. ISO 17025 is the relevant accreditation standard for analytical testing. A CoA from an unnamed or non-verifiable lab is worth treating with scepticism.
Red flags on a CoA
- No lab name or an unnamed testing facility
- No batch number linking the document to the product
- Test date more than 12 to 18 months old without explanation
- Beta-glucan percentage missing from the document
- Heavy metals or microbial sections absent entirely
- Numbers that do not match what the label claims
- A document that looks designed rather than clinical — branded, marketing-heavy, no raw data
That last one matters: some brands produce documents that look like CoAs but are internal quality summaries with no named external lab and no raw data. A real CoA looks like a lab report, not a marketing document.
How to request a CoA from a brand
Most brands do not advertise their CoAs prominently. Here is where to look and what to do.
Check the website first
Some brands publish CoAs directly on their product pages. Real Mushrooms links to batch-specific CoAs from each product page. Proactive publication is itself a positive signal.
Check the FAQ or contact page
Brands that do not publish CoAs publicly sometimes provide them on request via a contact form or email. Look for a quality or testing section in their FAQ.
Email the brand directly
If the CoA is not publicly available, a direct email works. Keep the request specific. Here is a straightforward template:
Hi, I’m considering your [product name] and would like to see the most recent certificate of analysis showing beta-glucan content and third-party testing results. Could you provide that, along with the batch number it corresponds to? Thank you.
Specific is better than vague. Asking for the beta-glucan content and the batch number makes it harder for a brand to send you a generic document that does not tell you what you actually need to know.
How brands typically respond
Brands that take quality seriously will send the document quickly. Some will point you to a published link you missed.
Brands with weaker products tend to respond differently. Common patterns include: a long delay with no document attached, a generic response about their quality standards without any actual data, a document that appears to be an internal summary rather than an independent lab report, or no response at all.
None of these are definitive, but they are signals worth weighing.
What if a brand will not provide a CoA
Then you have your answer about how much they have invested in verifying what is in their product.
A brand that cannot or will not provide an independent certificate of analysis for a current batch is asking you to trust their label claims without any mechanism for verification. In a supplement category with a documented quality problem, that is not a reasonable ask.
Some smaller brands produce good products without systematic documentation. But there are brands in this category that have invested in transparency. In the absence of a CoA, start with those.
How to Read a Mushroom Supplement Label
The bottom line
A certificate of analysis is the closest thing to objective evidence you can get for a supplement. It is produced by an independent lab, tied to a specific batch, and shows you verified numbers rather than label claims.
Most consumers never ask. Once you start, you notice quickly which brands have nothing to hide. That distinction is more useful than any star rating.
See the framework applied: We compared Real Mushrooms, Host Defense, and Oriveda against all five quality criteria. Read the full brand comparison →
Frequently asked questions
Not every brand runs systematic third-party testing. Some test irregularly or rely on supplier documentation. Brands that test consistently and publish results are making a deliberate transparency choice worth factoring in.
Outright forgery is uncommon at any real scale. The practical concern is documents that look official but come from non-accredited internal labs or are outdated. A named, verifiable lab addresses most of this.
A CoA is batch-specific. For a regularly purchased supplement, a document from the current production year is appropriate. One from several years ago tells you nothing reliable about what is in the product today.
That is a meaningful discrepancy worth taking seriously. Labels are not required to list beta-glucan content, so technically there may be no false claim. But if a brand implies potency through marketing language and the CoA shows 2 percent beta-glucans, you have learned something important about the gap between presentation and reality.
How this article was put together
This guide is based on supplement industry testing practices, independent lab testing standards including ISO 17025 accreditation requirements, and analysis of how mushroom supplement brands handle transparency requests. For informational purposes only. Not medical advice.
Sources and further reading
Certificate of analysis standards for dietary supplements are informed by ISO 17025 laboratory accreditation requirements and NSF International supplement testing frameworks. Independent mushroom supplement testing has been published by Labdoor and the American Botanical Council. For beta-glucan methodology see the International Mushroom Standards published by the American Herbal Pharmacopoeia.