Most people assume that supplements sold in the United States have been tested and approved before they reach store shelves. That assumption is reasonable. It is also incorrect.
The FDA does regulate supplements. But the way it regulates them is very different from how it handles pharmaceutical drugs, and understanding that difference is one of the most useful things a consumer in this category can know.
Mushroom Supplements: What Works, What’s Misleading, and How to Buy Safely
The framework: DSHEA 1994
The Dietary Supplement Health and Education Act, passed in 1994, established the current regulatory framework for supplements in the United States. Under DSHEA, dietary supplements are legally distinct from both foods and pharmaceutical drugs.
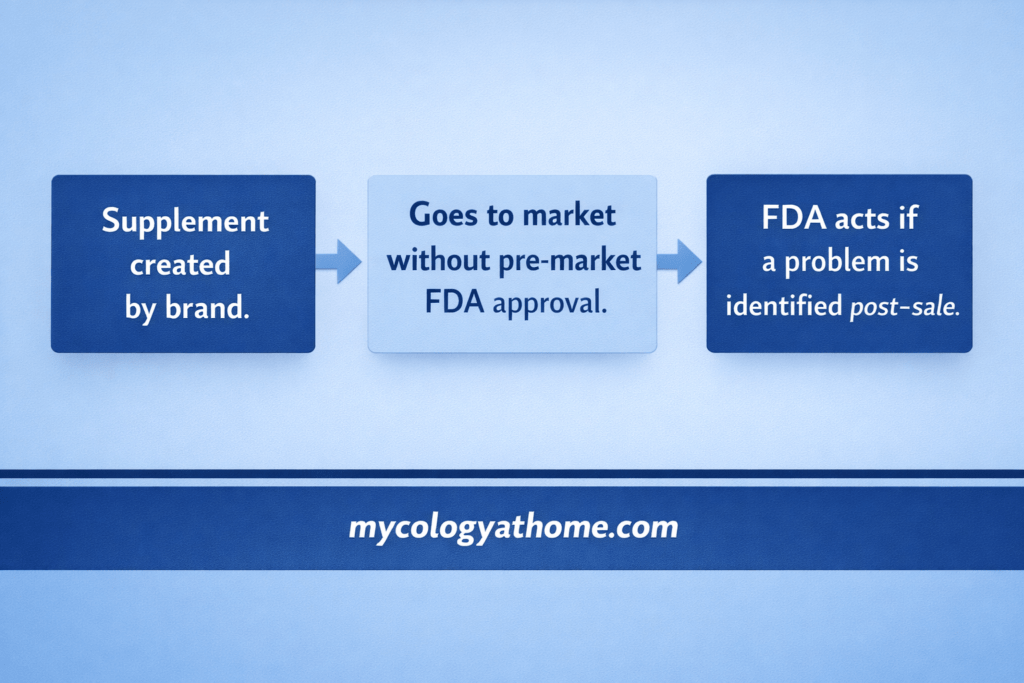
The most consequential part of DSHEA for consumers is this: a supplement company does not need FDA approval before bringing a product to market. The company is responsible for ensuring its product is safe and that its label is accurate, but the FDA does not review or verify this before the product goes on sale.
This is the opposite of the pharmaceutical model, where a manufacturer must demonstrate safety and efficacy through clinical trials before receiving approval. With supplements, the burden of proof is largely reversed.
What the FDA does regulate
DSHEA does not mean supplements are unregulated. The FDA has real authority in this category and exercises it in several ways.
Good Manufacturing Practices
Supplement manufacturers must follow FDA Good Manufacturing Practice regulations covering facility standards, equipment, personnel, and quality control. GMPs ensure consistent manufacturing, not verification of what is in a specific product, but they set a baseline.
Labelling requirements
Labels must include a Supplement Facts panel, ingredient list, manufacturer contact, and a disclaimer that the product has not been evaluated by the FDA. Drug claims are prohibited. Structure or function claims, such as supporting immune health, are permitted with the disclaimer.
Post-market enforcement
If a supplement is found to be unsafe or misbranded after it enters the market, the FDA can issue warning letters, request recalls, or pursue legal action. It also monitors adverse event reports from consumers and healthcare providers.
New dietary ingredient notifications
For ingredients not marketed before 1994, manufacturers must notify the FDA 75 days before market entry with safety information. This does not affect most established mushroom species.
What the FDA does not do
This is the part that surprises most consumers.
- The FDA does not test supplement products before they go to market.
- The FDA does not verify that a label accurately reflects what is in the product.
- The FDA does not confirm that a mushroom supplement contains the beta-glucan percentage implied by its marketing.
- The FDA does not confirm whether a product uses fruiting body or mycelium on grain.
- The FDA does not verify extraction methods or ratios.
- The FDA does not approve mushroom supplements.
A brand can produce a product, list 1000mg of lion’s mane on the label, make structure or function claims about immune and cognitive support, and go to market without any government body independently verifying the product contains what the label implies. That is how the system currently works.
This does not mean the FDA has no role. It means the role is reactive rather than preventive. The agency acts when problems are identified, not before products reach consumers.
Why this matters specifically for mushrooms
The regulatory gap has consequences that are more significant for mushroom supplements than for many other categories.
The active compounds in medicinal mushrooms, beta-glucans, triterpenes, hericenones, are not detectable by sight, smell, or taste. A capsule containing 30 percent beta-glucans from a quality fruiting body extract looks and feels identical to a capsule containing 2 percent from a grain-diluted mycelium powder. Consumers have no sensory way to evaluate quality, and the FDA does not fill that verification gap.
A brand can produce a poorly documented product, label it with unverified claims, price it competitively, and rely on the consumer’s inability to verify the contents. The regulatory framework does not prevent this.
Many brands are not doing this. It explains why the quality problem is persistent and why consumer-led verification matters.
Mycelium on Grain: The Problem With Most Mushroom Supplements
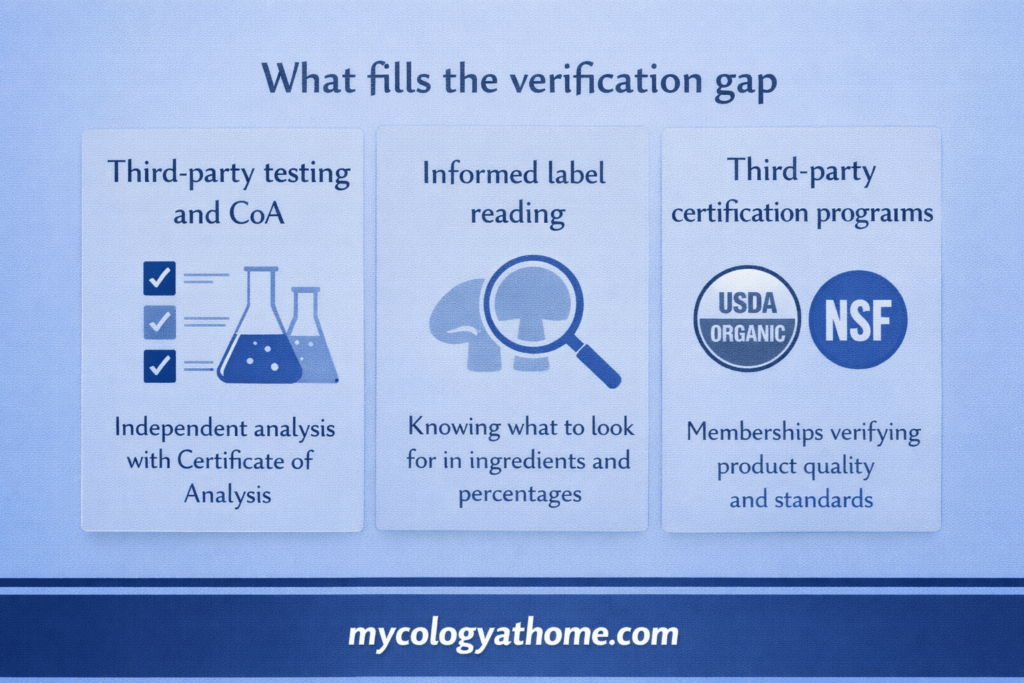
What fills the gap
Since pre-market verification by the FDA does not exist for this category, the tools available to consumers are third-party testing and brand transparency.
Third-party testing and certificates of analysis
Independent lab testing is the closest equivalent to pre-market verification available in the supplement market. A certificate of analysis from an accredited third-party lab verifies beta-glucan content, heavy metals, and microbial contamination for a specific batch. It is voluntary, which is why it serves as a useful signal of brand quality. Brands that invest in systematic independent testing and publish the results are demonstrating something the regulatory framework does not require.
What Is a Certificate of Analysis and How to Get One From a Mushroom Brand
Informed label reading
Understanding what supplement labels are required to show and what they are not gives you a much clearer view of what any given product is actually telling you. The gaps in a label are often as informative as what is present.
How to Read a Mushroom Supplement Label
Accreditation and certification programmes
Programs like NSF International, USP Verified, and Informed Sport verify manufacturing quality beyond GMPs through independent audits and product testing. Participation is voluntary but meaningful.
Common misconceptions worth clearing up
‘FDA approved’ supplements
No supplement sold under DSHEA is FDA approved. The term does not apply to this category. If a brand uses it, they are either misleading consumers or referring to something unrelated, such as the manufacturing facility.
‘Tested by the FDA’
The FDA tests supplements in response to specific complaints or enforcement actions, not as routine market oversight. A product that has not been recalled has not necessarily been tested.
‘Regulated means verified’
Regulation in the supplement category means manufacturing standards and post-market enforcement authority, not pre-market content verification. A regulated supplement can contain less than the label implies, use lower-quality material than the marketing suggests, and reach the market without any independent verification of its claims.
A simple rule of thumb
Regulation exists. It sets manufacturing standards and provides a mechanism for post-market enforcement. It does not verify the content of individual supplement products before they reach consumers.
In practical terms: FDA regulation means the product was likely made in a reasonably controlled environment. It does not mean the label accurately reflects what is in the capsule.
Brand transparency and independent testing are what fill the verification gap that regulation does not cover.
The bottom line
Understanding how FDA regulation of supplements actually works is not about distrust. It is about knowing how to evaluate quality in a framework where pre-market verification does not exist.
The system was designed to allow products to reach the market without the cost of pharmaceutical approval. That has benefits, but it also means consumers carry more verification responsibility than most people realise.
The good news is that once you understand the framework, the tools available, independent testing, certificates of analysis, and transparent labels, are sufficient to make well-informed decisions. You just need to know to use them.
Frequently asked questions
Is the FDA doing anything to improve supplement regulation?
There are ongoing discussions about mandatory product listing and enhanced adverse event reporting. The framework under DSHEA has remained largely intact since 1994 and changes move slowly.
Does a supplement manufactured in an FDA-registered facility mean it is verified?
Facility registration is a required administrative step, not a quality verification. It confirms the manufacturer has notified the FDA, not that the FDA has reviewed what they produce.
If a mushroom supplement passes GMP requirements, is it a quality product?
GMP compliance means consistent production under controlled conditions. It does not verify beta-glucan content, source material, or extraction method. Consistency of process and quality of output are different things.
What should I rely on if not FDA approval?
Third-party certificates of analysis, transparent labels specifying fruiting body and extraction method, and brands that publish testing data proactively. Those are the practical verification tools available.
How this article was put together
Based on DSHEA 1994, FDA guidance on dietary supplement regulation, and analysis of how the framework applies to the mushroom supplement category. For informational purposes only.
Sources and further reading
Regulatory information reflects DSHEA and subsequent FDA guidance. The FDA’s guidance on labelling, GMP requirements, and post-market enforcement is available at fda.gov.